Lenalidomide
Lenalidomide Impurities
High-purity reference standards for HPLC analysis, stability studies, and quality control.
Comprehensive Offerings
Alfa Omega Pharma provides certified reference substances, degradation products, and validated analytical methods fully compliant with pharmacopeial guidelines (USP, EP, ICH).
Custom Synthesis
We specialize in the custom synthesis of Impurities, Metabolites, and stable-labelled isotopes tailored to your specific research and regulatory needs.
Key Available Impurities & Derivatives:
- 4-Nitro Lenalidomide
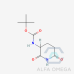
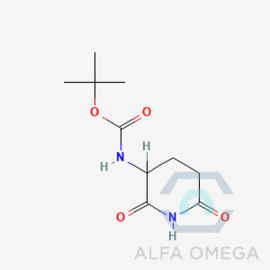
- tert-butyl (2,6-dioxopiperidin-3- yl)carbamate (Lenalidomide LMDL-02)
- Lenalidomide WS
- Lenalidomide Impurity-2
- Lenalidomide Impurity-1/Diacid
- Lenalidomide Impurity-3
- Lenalidomide N-Hydroxy Impurity
- Lenalidomide-N-Formyl Impurity
- Lenalidomide impurity-4
- Lenalidomide impurity-7/ Diamide
- Lenalidomide Impurity-1 Dimethyl Ester Lenalidomide Impurityurity 20
- Lenalidomide Imp-2 methyl Ester Lenalidomide Impurity 21
- Lenalidomide Imp-3 methyl Ester Lenalidomide Impurity 22
- Lenalidomide Impurity 2 & 3 mixture
- Lenalidomide Lactosamine adduct
- N-Acetyl Lenalidomide/1376
- Lenalidomide Peak- 1/LENALIDOMIDE IMPURITY C1(Z)
- Methyl 2-Methyl-4-nitrobenzoate
- Lenalidomide Impurity 16
- Lenalidomide Impurity 17
- Lenalidomide Impurity 18
- Lenalidomide Nitroso Impurity
- Lenalidomide Impurity-5
- Lenalidomide Peak- 2/LENALIDOMIDE IMPURITY C1(E)
- Pomalidomide Impurity B
From Discovery to delivery, High purity certified impurity standards.
Grid View:
tert-butyl (2,6-dioxopiperidin-3- yl)carbamate (Lenalidomide LMDL-02)
Product Details Overview Shipping Storage Downloads tert-butyl (2,6-dioxopiperid..